Description
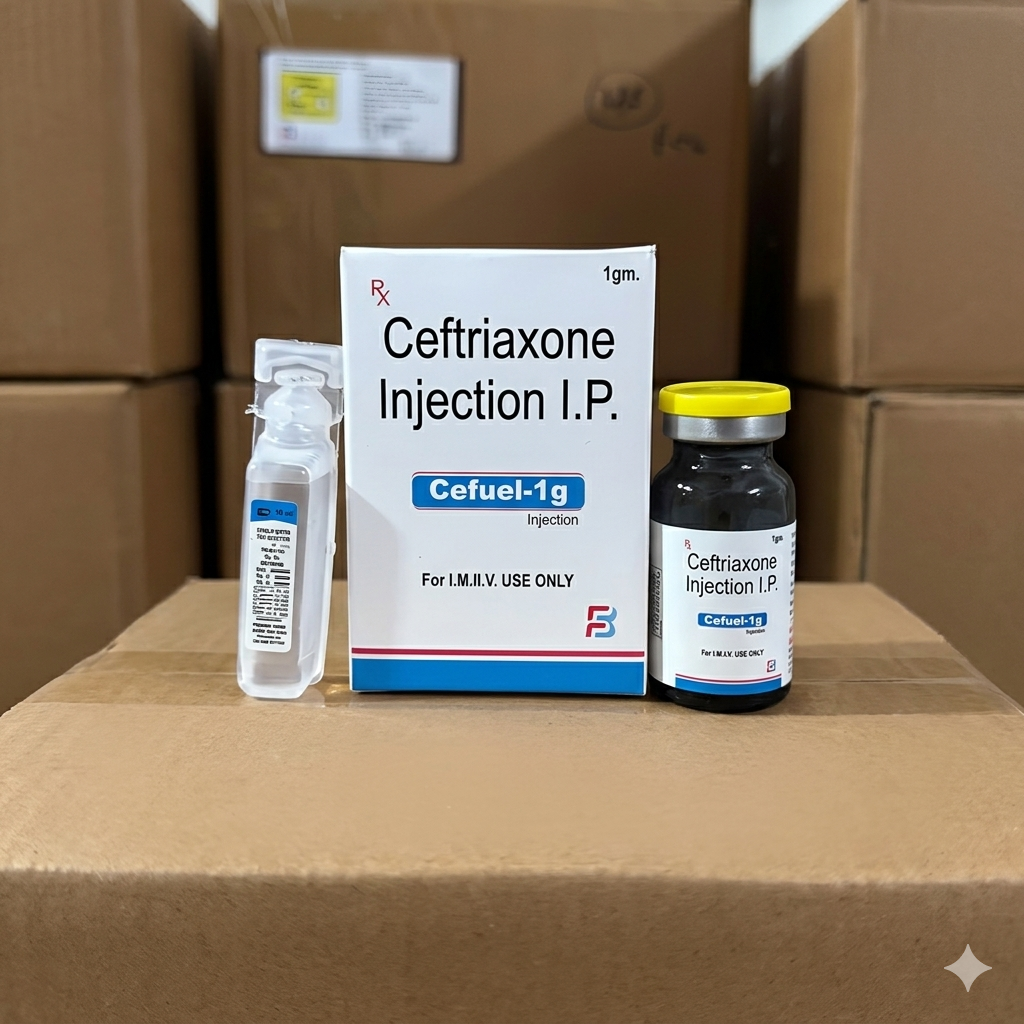
Cefuel-1g is a potent, broad-spectrum antibiotic used to treat a wide variety of bacterial infections. It contains Ceftriaxone, which belongs to the third-generation cephalosporin class of antibiotics. It is administered via injection and is effective against both Gram-positive and Gram-negative bacteria.
As indicated by the “I.P.” (Indian Pharmacopoeia) label, it is manufactured according to standardized pharmaceutical quality guidelines.
Composition
The medication consists of a sterile powder and a solvent for reconstitution:
-
Active Ingredient: Ceftriaxone Sodium I.P. equivalent to anhydrous Ceftriaxone: 1 gm.
-
Accompaniment: The package typically includes a sterile ampoule of Sterile Water for Injection (as seen in the clear plastic vial) used to dissolve the powder before administration.
-
Mechanism of Action: It works by interfering with the bacterial cell wall synthesis, leading to the eventual death of the bacteria.
Product Details
| Feature | Specification |
| Brand Name | Cefuel-1g |
| Generic Name | Ceftriaxone Injection I.P. |
| Strength | 1 gm |
| Route of Administration | For I.M. / I.V. Use ONLY (Intramuscular or Intravenous) |
| Legal Status | Prescription Only ($R_x$) |
Common Therapeutic Uses
Cefuel-1g is used by healthcare professionals to treat severe infections, including:
-
Respiratory Tract Infections: Such as severe pneumonia.
-
Meningitis: Infection of the membranes covering the brain and spinal cord.
-
Urinary Tract Infections (UTI): Complicated infections of the kidneys or bladder.
-
Bloodstream Infections: Such as sepsis or septicaemia.
-
Surgical Prophylaxis: Given before surgery to prevent post-operative infections.
-
Gonorrhea and other pelvic inflammatory diseases.
Important Safety Information
-
Professional Administration: This medication must never be self-administered. It should only be given by a doctor or a qualified nurse in a clinical setting.
-
Allergy Warning: Individuals with a known allergy to penicillins or cephalosporins must inform their doctor, as cross-reactivity can occur, leading to severe allergic reactions (anaphylaxis).
-
Reconstitution: Once the powder is mixed with the sterile water, it should be used immediately or stored according to the manufacturer’s specific instructions regarding stability.
-
Storage: The unmixed vial should be stored in a cool, dry place, protected from direct light.
Warning: Complete the full course as prescribed by your doctor, even if symptoms improve early, to prevent the development of antibiotic resistance.

Reviews
There are no reviews yet.